Влияние на аспекты ВИЧ-инфекции излечения от гепатита С у ко-инфицированных
-
Существуют противоречивые данные о влиянии вируса гепатита С (HCV) на иммунную активацию и воспаление у пациентов, коинфицированных вирусом иммунодефицита человека (ВИЧ) и HCV.
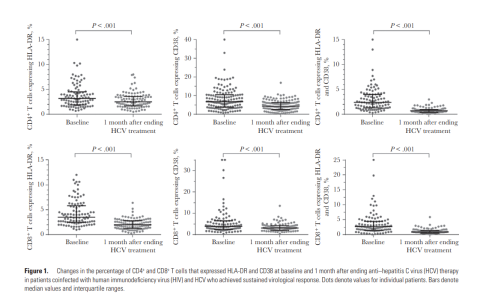
ВИЧ / HCV-коинфицированные пациенты, получающие антиретровирусную терапию, которые добились устойчивого вирусологического ответа с помощью безинтерфероновых режимов , последовательно регистрировались в проспективном исследовании. Следующие факторы были оценены до, сразу после окончания и через 1 месяц после окончания терапии: экспрессия HLA-DR / CD38, PD-1 и CD57 на CD4 + и CD8 + Т-клетках; измерение общей нагрузки ДНК ДНК в мононуклеарных клетках периферической крови; и определение уровней плазмы растворимого CD14 (sCD14), липополисахарида (LPS), 16S рибосомной ДНК (rDNA), интерлейкина 6 (IL-6), D-димеров и высокочувствительного C-реактивного белка (hsCRP).
Подробнее:
https://academic.oup.com/jid/article-abstract/218/4/624/5050122?redirectedFrom=fulltext
-
@васьвась ну, очень примерно так.
Визуально данная история:
И как-то так:
The fact that HCV coinfection induces a higher level of immune activation in HIV-infected patients was inferred from comparison of immune activation levels in monoinfected and coinfected patients. Only the study by Gonzalez et al [17] has shown a decreased frequency of CD38 expression on CD8+ T cells among patients (n = 14) who achieved SVR. Our study assessed the medium-term (ie, 1 month after HCV treatment cessation) impact of HCV eradication due to interferon-free regimens on several parameters related to immune activation and immune recovery in a large group of HIV/HCV-coinfected patients.
In addition to a significant reduction in CD38 and HLA-DR expression on CD4+ and CD8+ T cells, there were decreases in levels of proviral HIV DNA, sCD14, microbial translocation markers, and D-dimers after the eradication of HCV infection. Although there were not quantitative correlations between the decrease in proviral HIV DNA load and the expression of HLA-DR and CD38 on both CD4+ and CD8+ T cells, it might be hypothesized that both events might be related. In HIV/HCVcoinfected patients, HCV eradication has proven to be beneficial not only in terms of the liver-related mortality rate and fibrosis regression, but also with regard to decreases in the frequencies of HIV disease progression, non–AIDS-related events, and non–liver-related mortality [26–30]. Almost certainly, a large proportion of this benefit is due to the reduction in the level of immune activation, given its key role in driving HIV-1 disease and non–AIDS-defining events in this population [31, 32]. By contrast, the expression of immune dysfunction (PD-1) and immune senescence (CD57) markers on both CD4+ and CD8+ T cells, as well as the IL-6 and hs-CRP plasma levels, did not change after SVR, suggesting that HCV has a limited or null influence on them in HIV/HCV-coinfected patients.
On the other hand, based on the adverse effect that HCV has on immune recovery among HIV-infected patients initiating ART [33–35], we hoped that the eradication of HCV would contribute to restoring immunologic parameters in these patients. However, the achievement of SVR did not translate into an increased CD4+ T-cell count, CD4+ T-cell percentage, or CD4+/ CD8+ T-cell ratio. In this regard, a limitation of our study may be the limited number of patients with very low CD4+ T-cell counts and CD4+/CD8+ T-cell ratios included in the study. It would be necessary to evaluate a larger sample of such patients to state with confidence that improvement in these parameters should not be expected in very immunosuppressed HIVinfected patients after the eradication of HCV. However, this is in concordance with results of a previous retrospective study by Saracino et al [29], in which SVR did not seem to alter the rate of long-term improvements in the CD4+ T-cell count and CD4+/ CD8+ T-cell ratio in HIV/HCV-coinfected patients receiving ART. This is particularly worrying in HIV-infected patients who did not achieve significant immune recovery despite consistent HIV suppression, since they have shown higher rates of morbidity and mortality due to AIDS-defining and non–AIDSdefining events [36].
Our study has other limitations, one of which is that the follow-up period was limited to 1 month after finishing HCV treatment. Data from a longer follow-up period would have reinforced the conclusions. However, the concordance between values at the end of treatment and those 1 month later allows us to be confident that these findings are immunologically relevant. Likewise, the inclusion of patients who did not achieve SVR would be very informative, but it would currently be very difficult to do so because of the elevated rate of response to the direct-acting antiviral agents.
In conclusion, our data suggest that the eradication of HCV infection in HIV/HCV-coinfected patients results in significant decreases not only in immune activation of CD4+ and CD8+ T cells, but also in levels of proviral HIV DNA, microbial translocation markers, sCD14, and D-dimers. It supports the use of HCV treatment in all HIV/HCV-coinfected patients regardless of fibrosis stage, with the sole exception of decompensated cirrhosis.
-
@ilya-antipin ура самоизлечение,у меня качественный на гепс отрицательный)))
-
@ilya-antipin илья а скажите амегу от фирмы"" solgar""какую лучше?есть 3-6-9 в комплексе а есть просто омега3
-
@guf Омегу. Через О. Чтобы сказать, что лучше – должны быть жесткие ориентиры. А их нет.
Мы рассматриваем Омега3 как что? Нам состояние мембран нейрончиков улучшить? Липидный профиль, что-то с триглицеридами? Или же мы думаем о воздействии на системное воспаление? Чтобы что? Отсюда танцевать нужно.
А может просто семгу чаще покупать? Я убежден, что сперва нужно вывести потребление морской рыбы минимум на 30 кило в год, а после этого уже думать о всяких капсулках. -
@ilya-antipin для общего укрепления здоровья омегу можно пропить?чисто для профилактики всего что вы перечислили!
-
@ilya-antipin стейки из лосося тоже буду кушать хотябы раз в недельку)))
-
@guf наверное, но такая мотивация мне не очень понятна. В таком случае можно весьма произвольно принимать, т.к. опять же ориентиров то ноль. «Тебе что?» «Молоток» «Для чего? Какой? Забивать шлямбура в скалу, гвоздики в ботинки, работать на крышах, или мясо отбивать, или для жестчнки в автосервисе» «Вот, для всего, что перечили. Просто, чтобы был»… ну, вот так это не работает.
-
@ilya-antipin я слышал что омега3 полезна для сердечно сосудистой системы и для мозга,темболее схема ваарт у меня на основе ип "дарунавир"ещё где-то слышал что ип могут повышать уровень холестерина,+к этому я стараюсь держать себя в форме и отжимаюсь по 50 раз с непривычки но дальше больше,и качаю прес,считаю что омега мне необходима для набора мышечной массы и ссс.
-
-
я слышал что омега3 полезна для сердечно сосудистой системы
Только если выраженный дефицит, если боле или менее поступает с пищей, то не доказаны выгоды. За исключением ситуации с триглицеридами – может снижать их уровень, опять же, если там есть что-то.
и для мозга
Улучшает параметры мембран некоторые, клиническая значимость этого пока под вопросом.
Вообще понятие полезно не есть понятие медицинское. Нет в медицине ничего полезного. Есть что-то, что с вероятностью такой-то при нозологии такой-то приводит с вероятностью такой-то к таким эффектам при такой вот безопасности. Это и есть лекарства. Вы не о них.
-
@васьвась К теме - почитала данные исследования и интересно стало проверить на себе - работает :) После лечения гепатита С вирусная нагрузка со 130 000 упала до 81 000 (без терапии). Разница на 50 000 все таки существенная, на СD4 не повлияло, СD8 упал с 1341 до 802. ИРИ с 0.43 поднялся до 0.58
-
@senya_s было плохо, а стало плохо. Хм… прогресс.
-
@ilya-antipin А я и не утверждала что все стало гораздо лучше, просто отметила разницу ВН.
-
Участник @senya_s написал в Излечение гепатита С снижает иммунную активацию, микробную транслокацию и уровень ДНК ВИЧ у пациентов с коинфекцией ВИЧ / HCV:
просто отметила разницу ВН.
А причем тут гепатит? Ну скачет ВН туда-сюда, обычное дело.
У тебя миллиарды вирионов разрушают организм сейчас. Что в этом хорошего? -
@senya_s нет разницы.

